How to Get a BIS Certificate
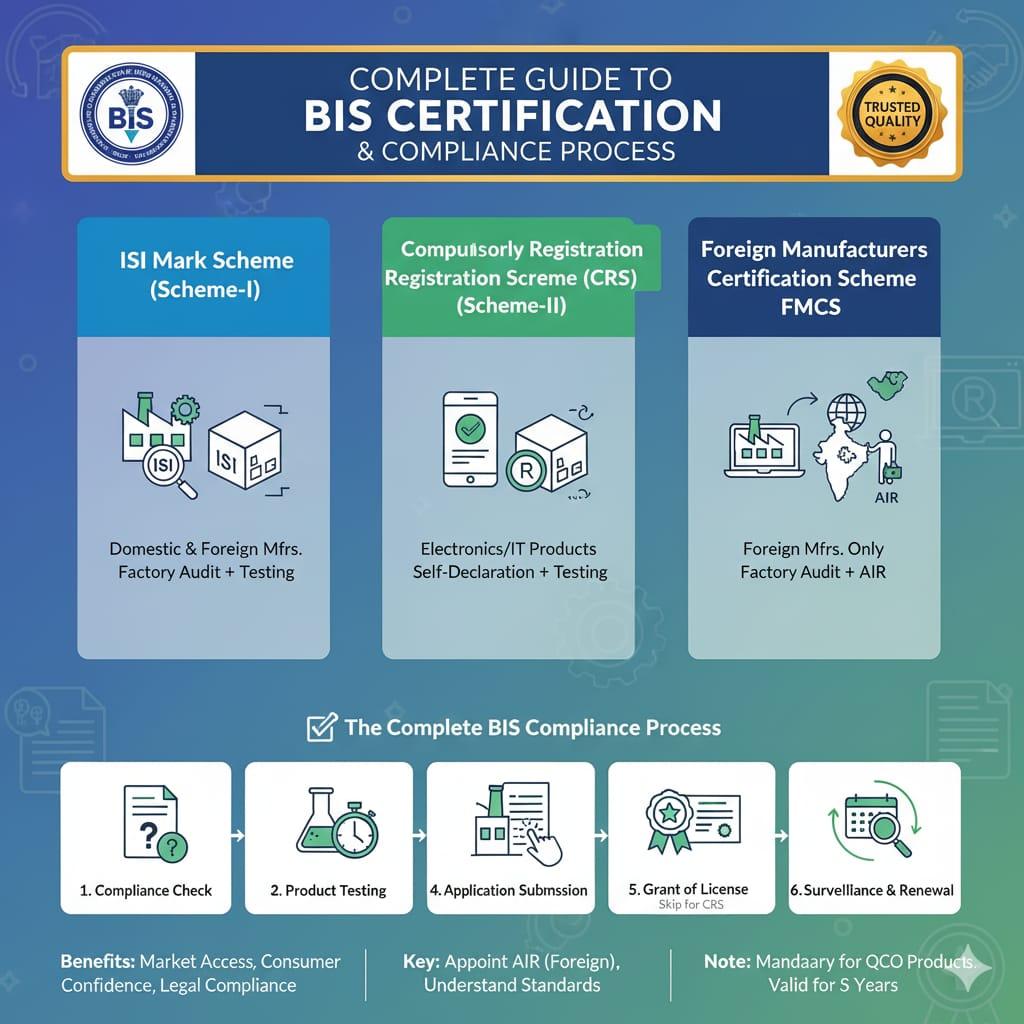
The process of obtaining a BIS Certificate starts with identifying the correct certification route for your product. First, confirm whether your product is covered under the ISI Certification Scheme, which involves licensing, factory surveillance, and use of the ISI mark, or under the Compulsory Registration Scheme (CRS), which mainly applies to electronics and IT products. Once the scheme is identified, determine the applicable Indian Standard (IS) and carefully review its scope. The scope specifies mandatory tests, labeling and marking requirements, and any exclusions.
Next, prepare a structured compliance plan. List all product models and variants, map the relevant clauses of the standard to each product, and collect the required technical and regulatory documents. These typically include product drawings and specifications, a list of critical components, safety declarations, and evidence of a quality management system (QMS). Proper planning at this stage helps prevent delays during testing and application review.
Product testing must be carried out at a BIS-recognized laboratory with experience in your product category. Finalize the number of samples, testing timelines, and the required report format before testing begins. While testing is in progress, compile your application dossier, including business registration documents, brand authorization (if applicable), labeling or marking artwork clearly referencing the relevant IS, and—if the manufacturer is located outside India—the appointment of an Authorized Indian Representative (AIR).
Under the CRS route, once the product successfully passes all tests, the test reports and supporting documents are submitted online through the BIS portal. After payment of the applicable fees and approval by BIS, a Registration Number (R-number) is issued.
For products covered under the ISI Scheme, BIS conducts a factory inspection to assess production controls, in-house testing facilities, and marking procedures. After the license is granted, ongoing surveillance and periodic audits are conducted to ensure continued compliance.
After certification, manufacturers must strictly follow BIS marking and packaging rules, maintain production and test records, and inform BIS of any changes in components, firmware, suppliers, or design. Establishing a renewal schedule and a clear process for adding new variants helps keep the entire product portfolio continuously compliant.
URL:- https://bis-certifications.com/how-to-get-bis-certificate
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Παιχνίδια
- Gardening
- Health
- Κεντρική Σελίδα
- Literature
- Music
- Networking
- άλλο
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


